Adoptive T-Cell Transfer in Mice

Back to Pharmacology and Toxicology Assessment top page
Induction of Adoptive T-Cell Transfer in Mice:
On study day 0, spleens for naive T cell isolation are obtained from female donor mice using the SCID or Rag2 IBD cell separation protocol1. After cells have been obtained and sorted, each female recipient mouse receives an IP injection of a minimum 4×105 cells/mouse (200 µL/mouse injections).
Disease Parameters/Progression:
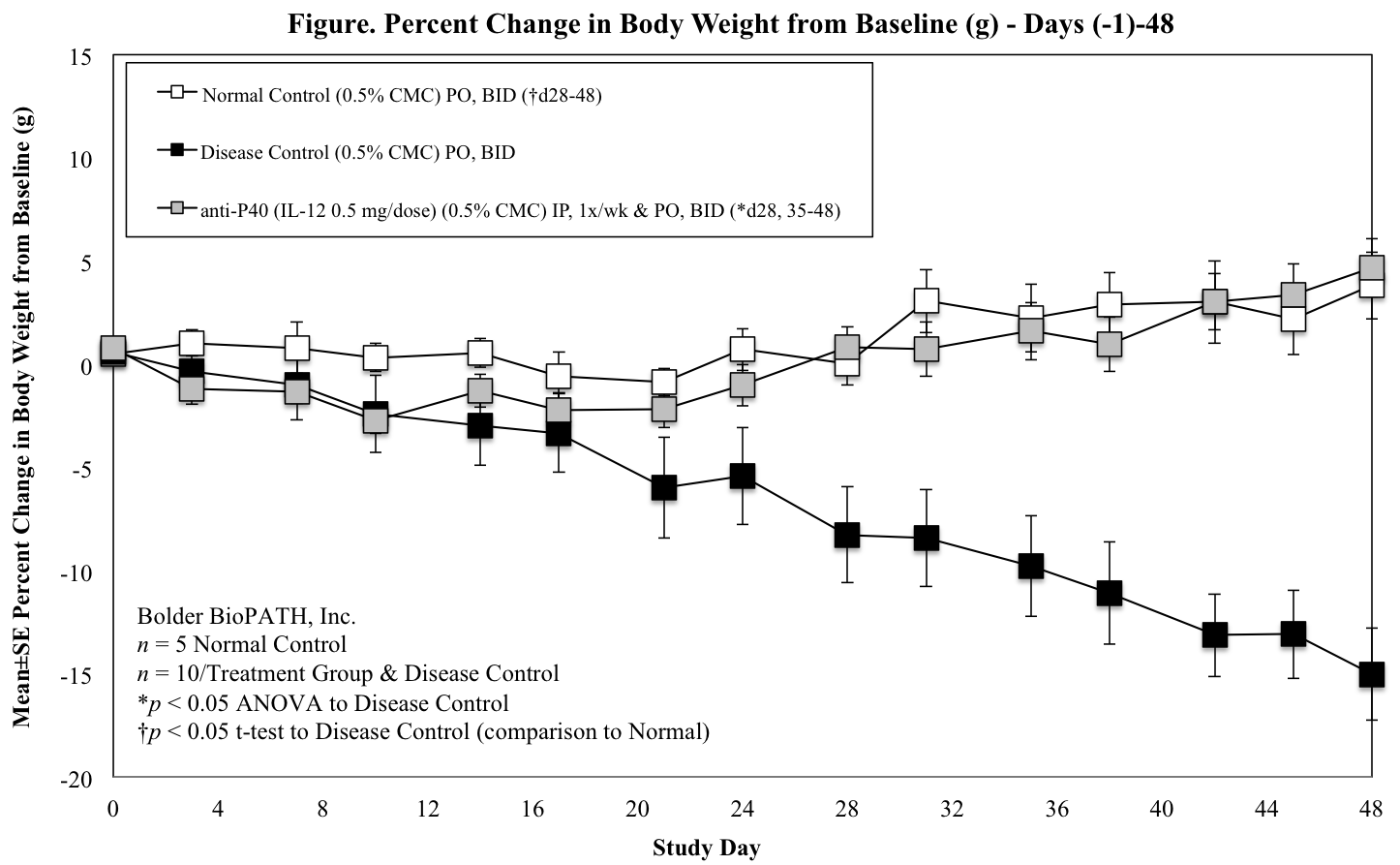
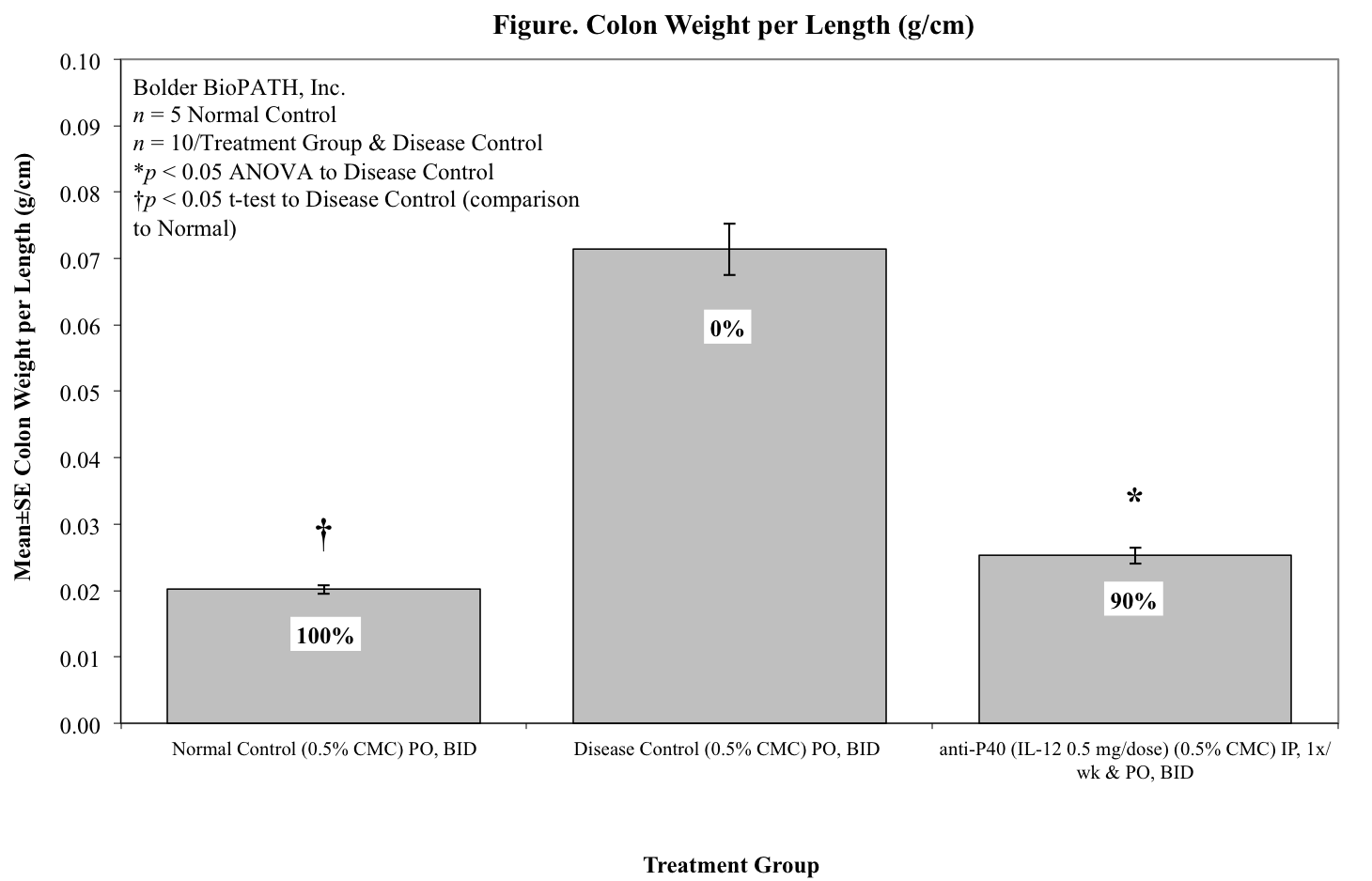
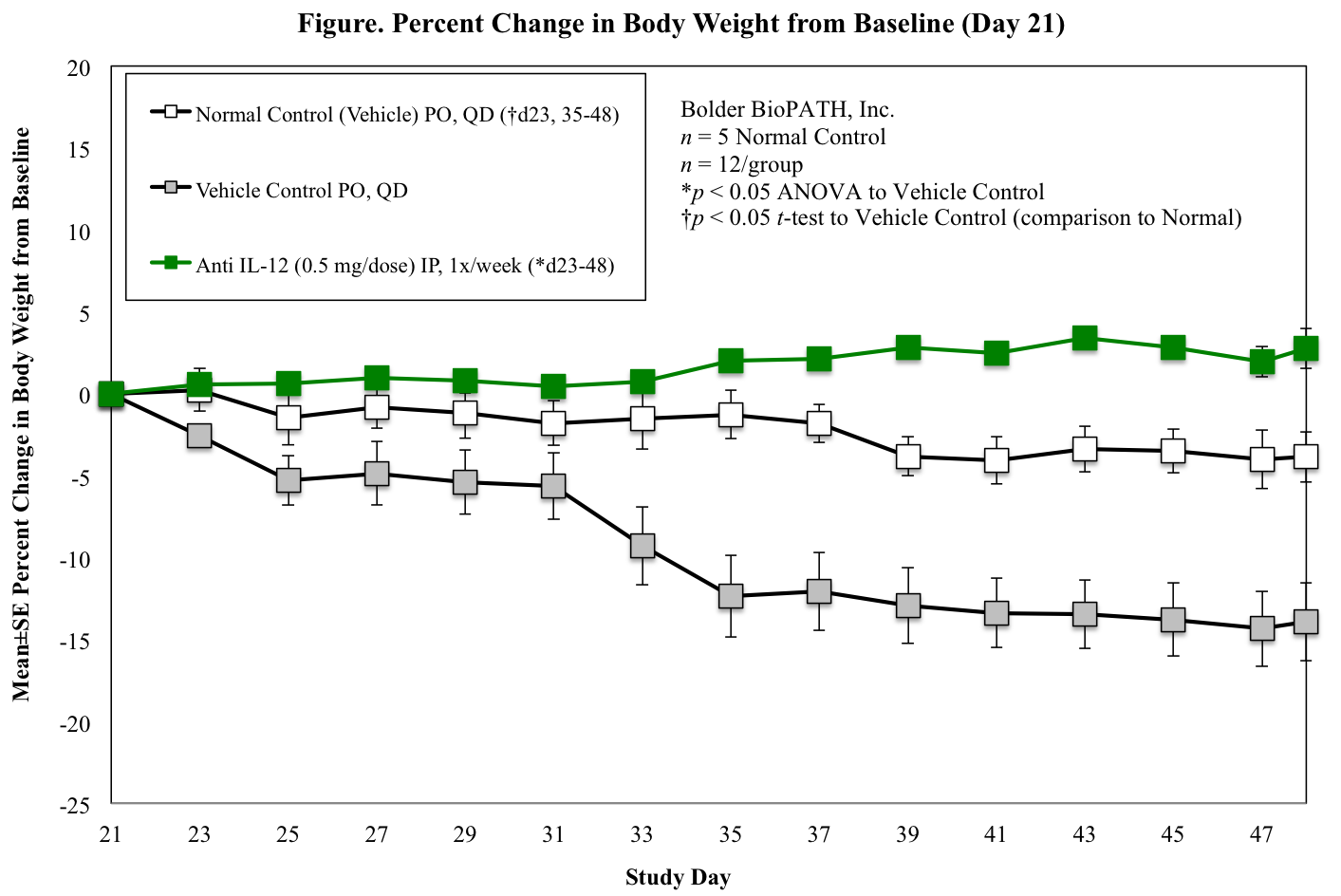
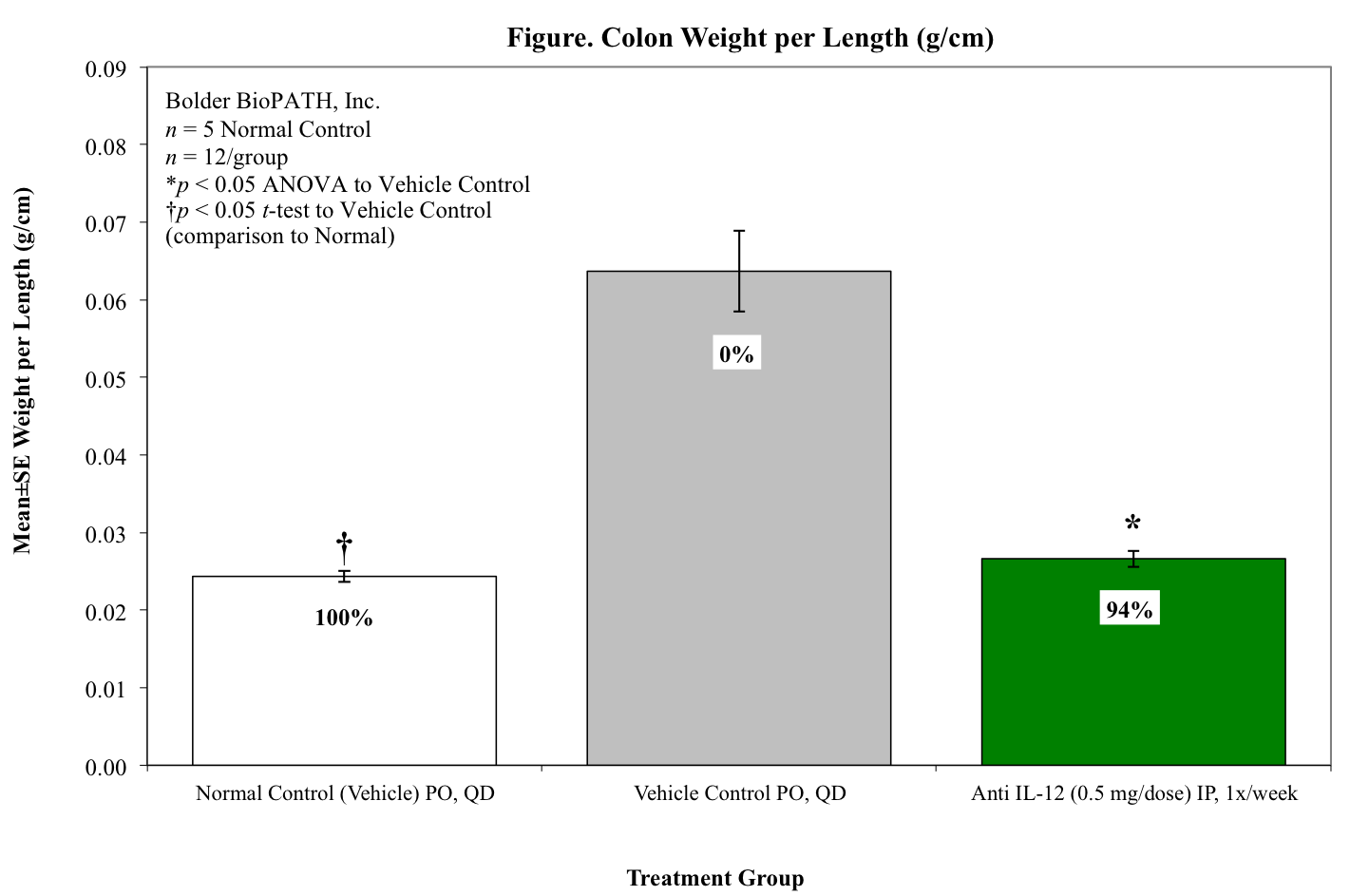
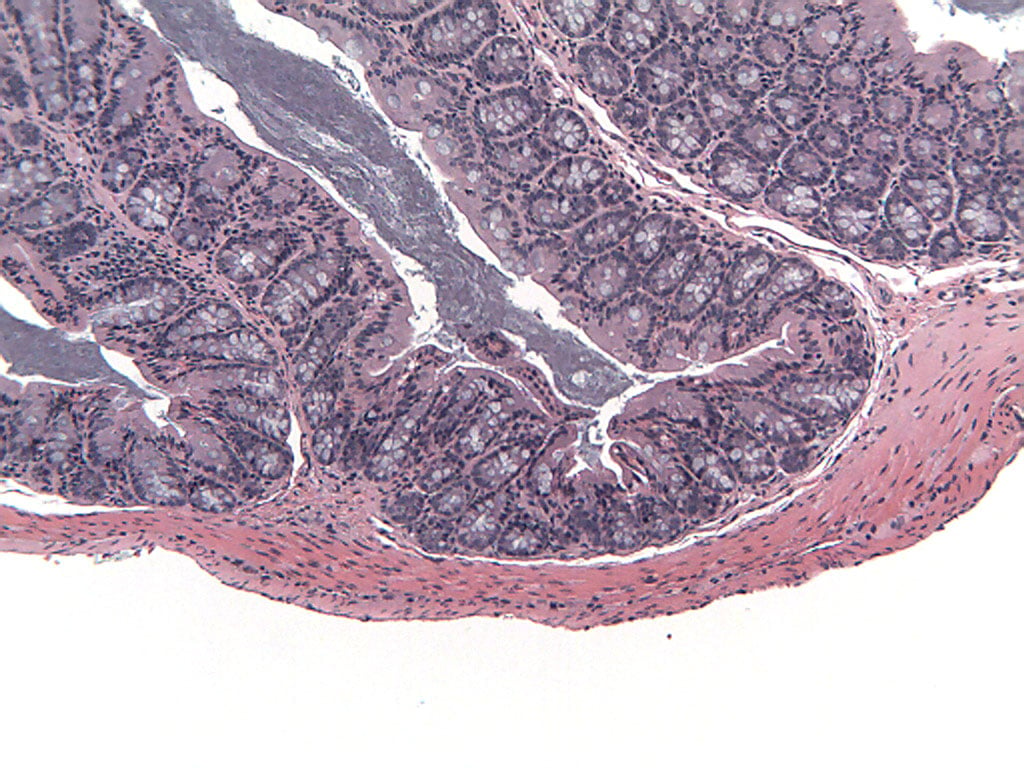
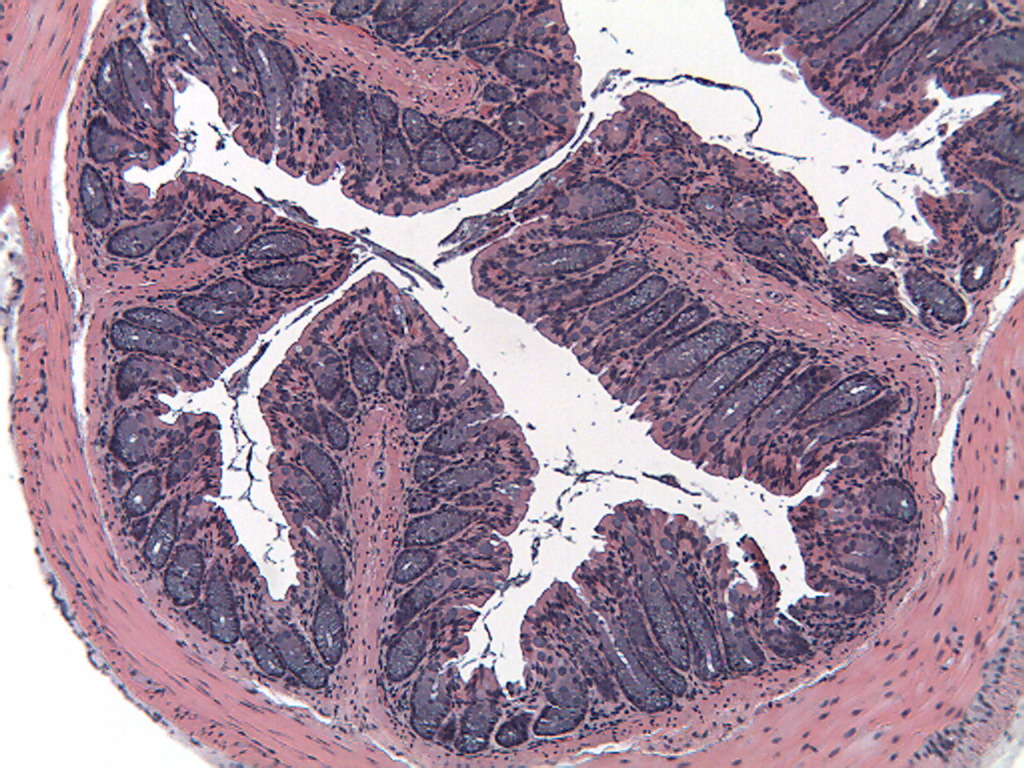
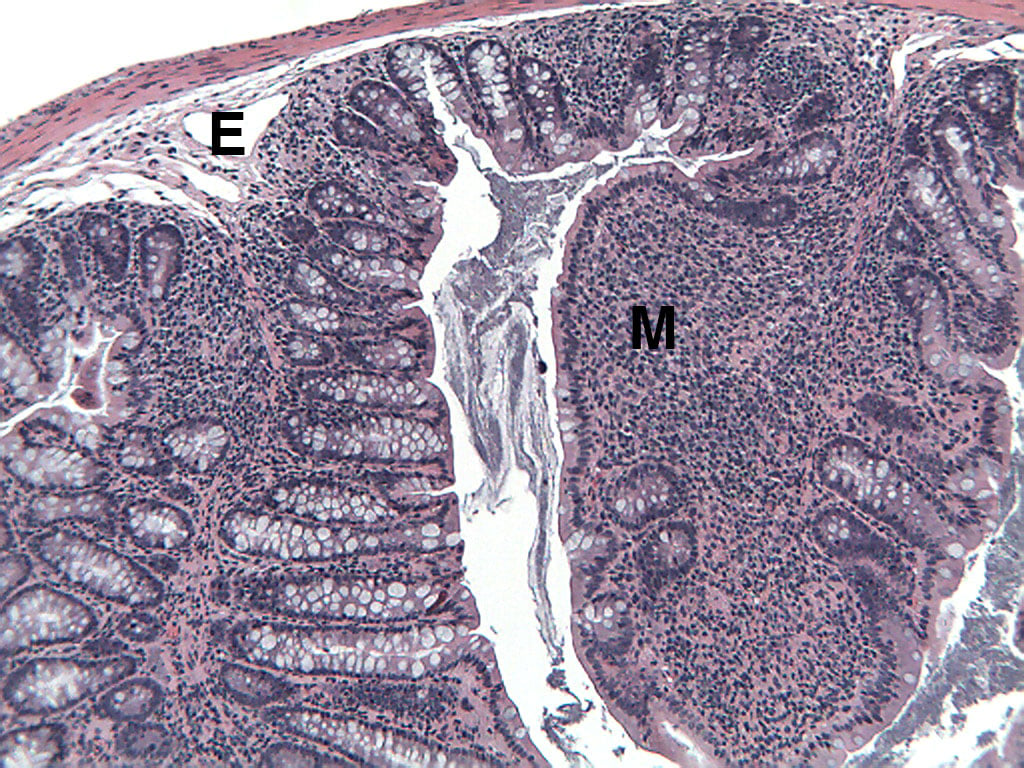
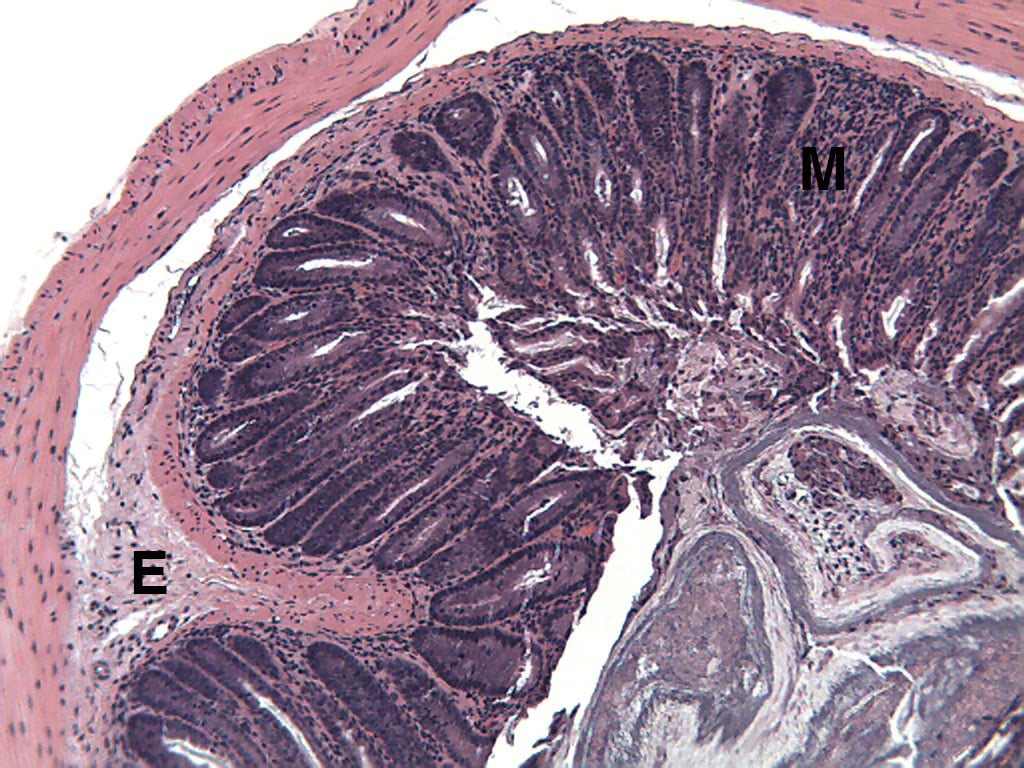
SCID or Rag2 mice restored with naive T cells start losing weight and developing lose stools 3 to 5 weeks after reconstitution. Weight loss is progressive, with mice losing up to 25% of body weight within 10 weeks of reconstitution. At necropsy, approximately 80 to 90% of mice have extended colons. The majority of SCID or Rag2 mice restored with naive T cells develop colitis with moderate to marked epithelial cell hyperplasia, significant to extensive leukocyte infiltrate in mucosa and submucosa, significant depletion of mucin-secreting goblet cells, and ulceration1. Inflammatory and epithelial lesions occur with the greatest incidence and severity between the transverse colon and the rectum2.
Dosing Paradigms:
- Prophylactic – Begin Dosing prior to or on the day of inoculation (Day 0) and continue until necropsy on day 49.
- Semi-established – Begin dosing on study day 14 and continue until necropsy on day 49.
- Established (Therapeutic) – Begin dosing on study day 21 and continue until necropsy on day 49.
- Route of administration: SC, PO, IP, IV, IC
Clinical Assessment:
Mice are weighed at least twice a week once dosing begins. At necropsy, the entire colon from each animal is harvested, inspected visually, measured for length, and weighed. Only animals that survived to study termination are included in the analysis of terminal colon lengths and weights.
Histopathological Assessment:
Colon sections are examined microscopically by a board certified veterinary pathologist (Dr. Alison Bendele) and scored (as proximal colon, distal colon, and total colon). Methods upon request.
Sample Data (Click on image to enlarge):
Mouse Clinical
Mouse Histopathology
Notes:
Transfer of naive T cells into the severe combined immunodeficiency (SCID) or recombination activating gene 2 (Rag2) deficient mouse models is a highly reproducible and easily manipulated model of colitis that has characteristics similar to chronic, idiopathic inflammatory bowel disease (IBD) in humans. Female Fox Chase C.B-17 SCID or Rag2 -/- mice, which lack B and T cells, are inoculated intraperitoneally (IP) with CD45RBhigh CD4+ T cells from congenic donor mice to induce disease with morphological features resembling human ulcerative colitis (UC) or Crohn’s disease (CD)1. IBD induced in SCID or Rag mice by transfer of CD45RBhigh CD4+ T cells involves differential activation of TH1 cells, which emphasizes the importance of an intact immune system in regulating intestinal inflammation2. Intestinal microflora also play a role in the development of the disease; however the mechanisms by which microflora influence mucosal immune responses are unclear3. In this model, mice develop intestinal lesions extending diffusely from the cecum to the rectum, which are characterized by extensive leukocytic infiltrates, epithelial cell hyperplasia with glandular elongation, ulceration, and loss of mucin-secreting goblet cells. Clinical symptoms include progressive weight loss and loose, mucous stool1. This model may be useful for studying mucosal immunoregulation as it relates to the pathogenesis of IBD. Immune manipulations that inhibit TH1 cell activation, such as anti-IFN-g monoclonal antibody treatment or systemic administration of recombinant IL-10, have been shown to be of significant benefit in this model1.
Optional Endpoint
- PK/PD blood collections
- Cytokine/chemokine analysis via Luminex(R)
- Other sandwich ELISAs
- CBC/clinical chemistry analysis
- Soft tissue collection
- Histopathologic analysis
- Immunohistochemistry analysis
- Endoscopy
References
- Read S, and Powrie F. Induction of Inflammatory Bowel Disease in Immunodeficient Mice by Depletion of Regulatory T Cells. Curr Protoc Immunol. 1999;30 suppl:15.13.1–15.13.10.
- Leach, Michael W., Andrew G. D. Bean, et al. Inflammatory Bowel Disease in C.B-17 SCID Mice Reconstituted with the CD45RBhigh Subset of CD4+ T Cells. Am J Pathol. 1996;148(5):1503–1515.
- Boismenu R, and Chen Y. Insights from mouse models of colitis. J Leukoc Biol. 2000;67(3):267–278.
For more information about CD45RBhigh Adoptive T-Cell Transfer (Mouse) contact us here.