Bleomycin-Induced Scleroderma in Mice

Induction:
Systemic sclerosis (SSc) is an autoimmune connective tissue disorder that is varied in its clinical manifestations but is characterized by circulating autoantibodies, vasculopathy, and fibrosis of the skin, lung, kidney, and GI tract1-6. Due to the accumulation of collagen, skin symptoms and stiffness may cause severe disability, while cardiovascular and pulmonary complication lead to high mortality1,2,7. Despite recent progress and numerous efforts to understand the underlying mechanisms of the disease, therapies are limited.
The lack of an established therapy for SSc is hindered by the lack of animal models that fully recapitulate the complex nature of this disease7,8. Although no animal model currently reproduces all the manifestations of SSc, the bleomycin-induced scleroderma model is the most widely used and well characterized. Because of its abilities to cleave DNA, bleomycin (BLM) is an anti-tumor antibiotic used in various cancers, which was originally isolated from the fungus Streptomyces virticullus5,8. While the exact mechanism of BLM-induced SSc is not yet fully elucidated, it is hypothesized that BLM causes dermal injury which activates inflammatory cells such as macrophages, T-cells, neutrophils, eosinophils, and mast cells which leads to apoptosis, reactive oxygen species (ROS), and a cytokine and chemokine cascade leading to fibroblasts and endothelial cells increasing production of the extracellular matrix (ECM), ultimately resulting in scleroderma2,3,5,7,9,10. The bleomycin-induced model reflects many of the symptoms seen in patients, particularly circulating autoantibodies and fibrosis of the skin and lung1-10.
Disease Parameters:
On study day -1 animals will be anesthetized and the hair on the back will be depilated. Mice may be re-shaved as necessary to keep the back free of hair.
On study day 0, animals will receive daily SC 100ml injections of 1mg/ml bleomycin or vehicle for four weeks; alternatively, animals may be administered bleomycin via subcutaneous osmotic minipump (Alzet Pump) or nasal application. Minipumps will be filled with up to 27mg/ml bleomycin or vehicle and kept implanted for four weeks. Some animals may be administered 50ml of 3mg/ml bleomycin once a week for four weeks intranasally to increase lung involvement. For intranasal administration, animals will be briefly anesthetized with 3% isoflurane and held in a vertical position wile 50ml of BLM or vehicle is instilled into the nose via a pipette. Animals will be kept in an upright position for at least 2 minutes to allow the fluid to trickle into the lungs. Afterwards, animals will be returned to their home cage for recovery.
Animals may be necropsied up to two weeks after the final dose of bleomycin.
Dosing Paradigms:
- Developing (Prophylactic) – Begin dosing on study day 0 and continue until necropsy on day 28.
- Semi-Established (Prophylactic) – Begin dosing between study days 7-14 and continue until necropsy on day 28.
- Established (Therapeutic) – Begin dosing after study day 14 and continue until necropsy on day 28.
Endpoints
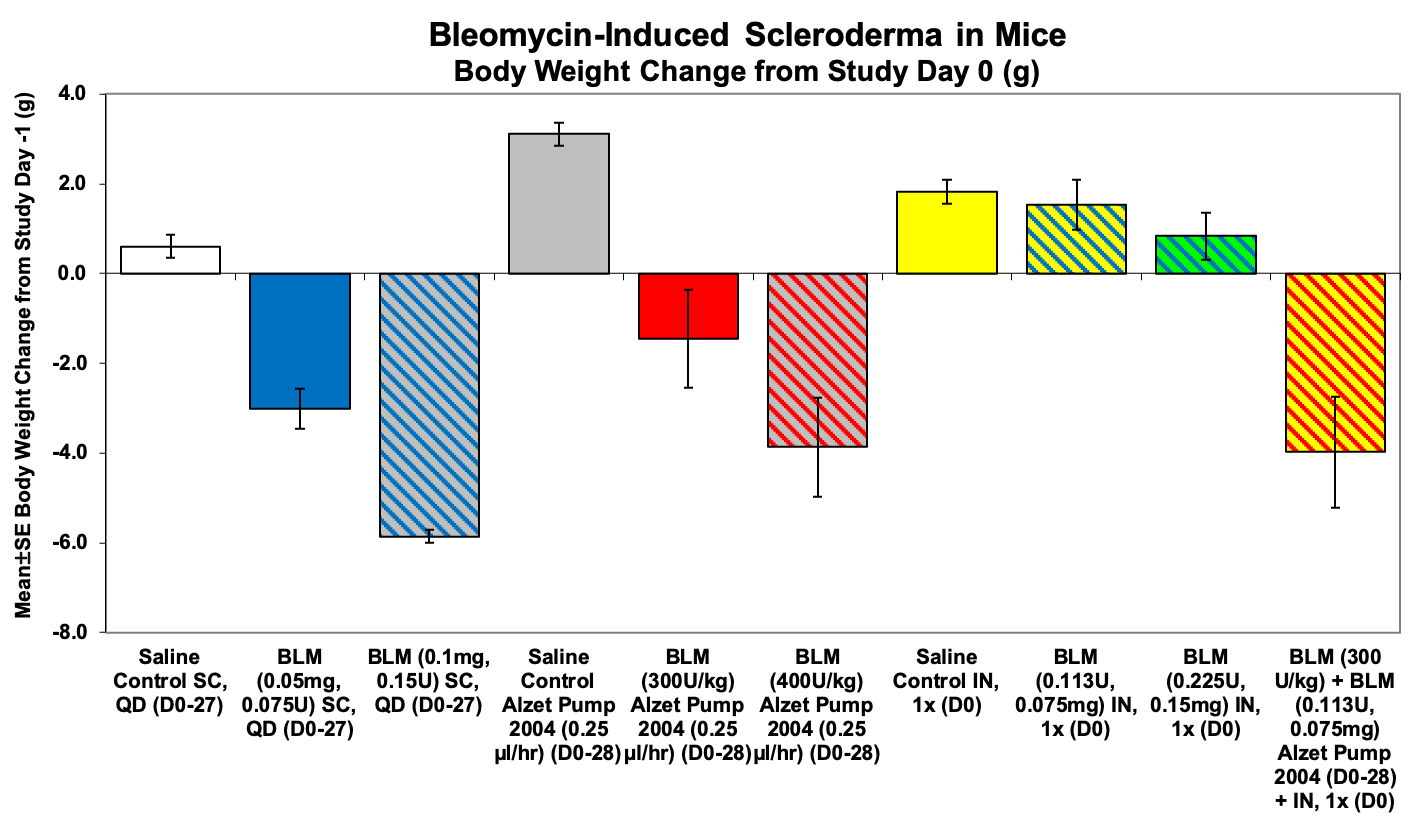
-Body weight
-Skin punch weight
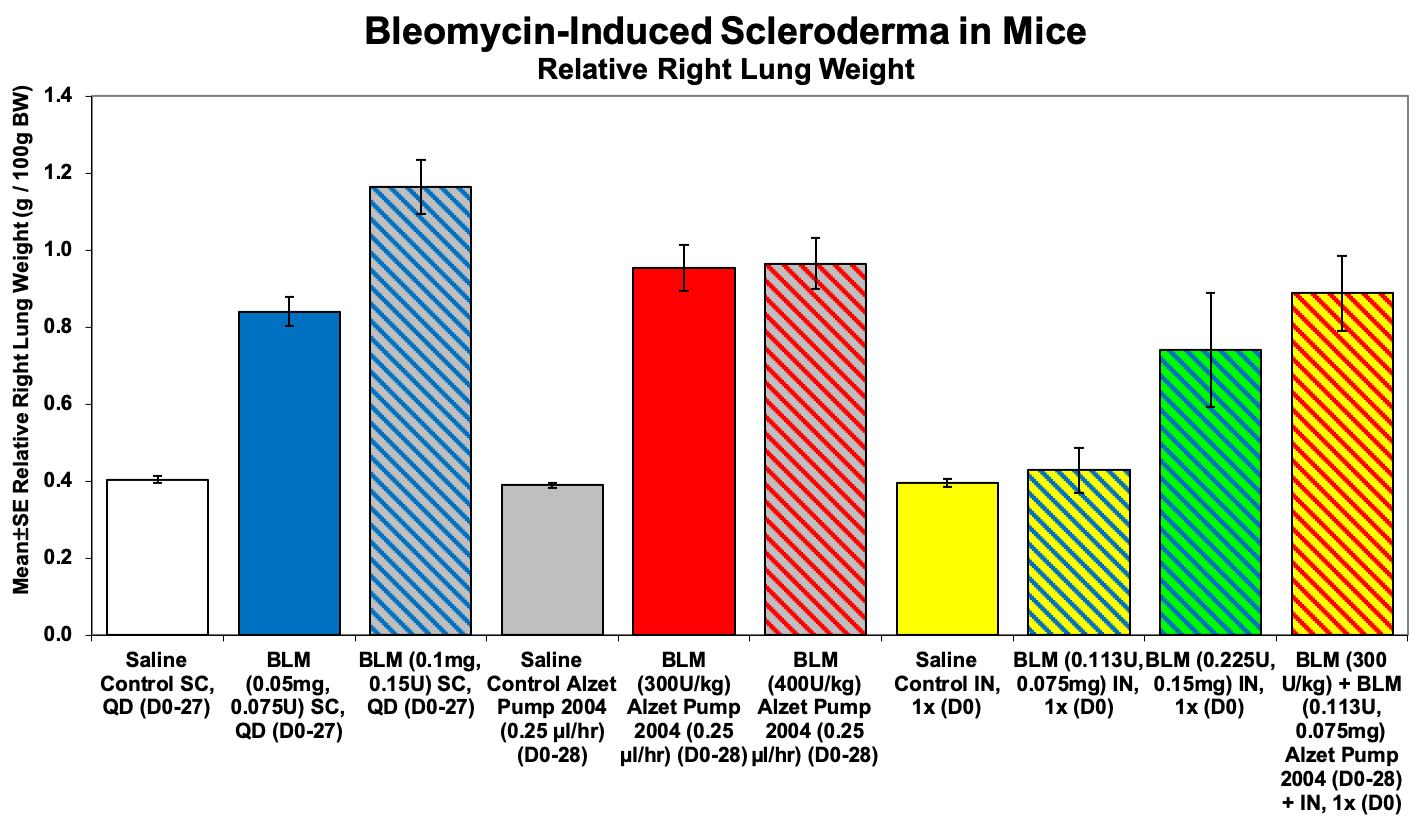
-Lung weight
-Skin histopathology
-Lung histopathology
-ELISA and/or Luminex analysis of serum, skin, and lung homogenate
Histopathological Assessment:
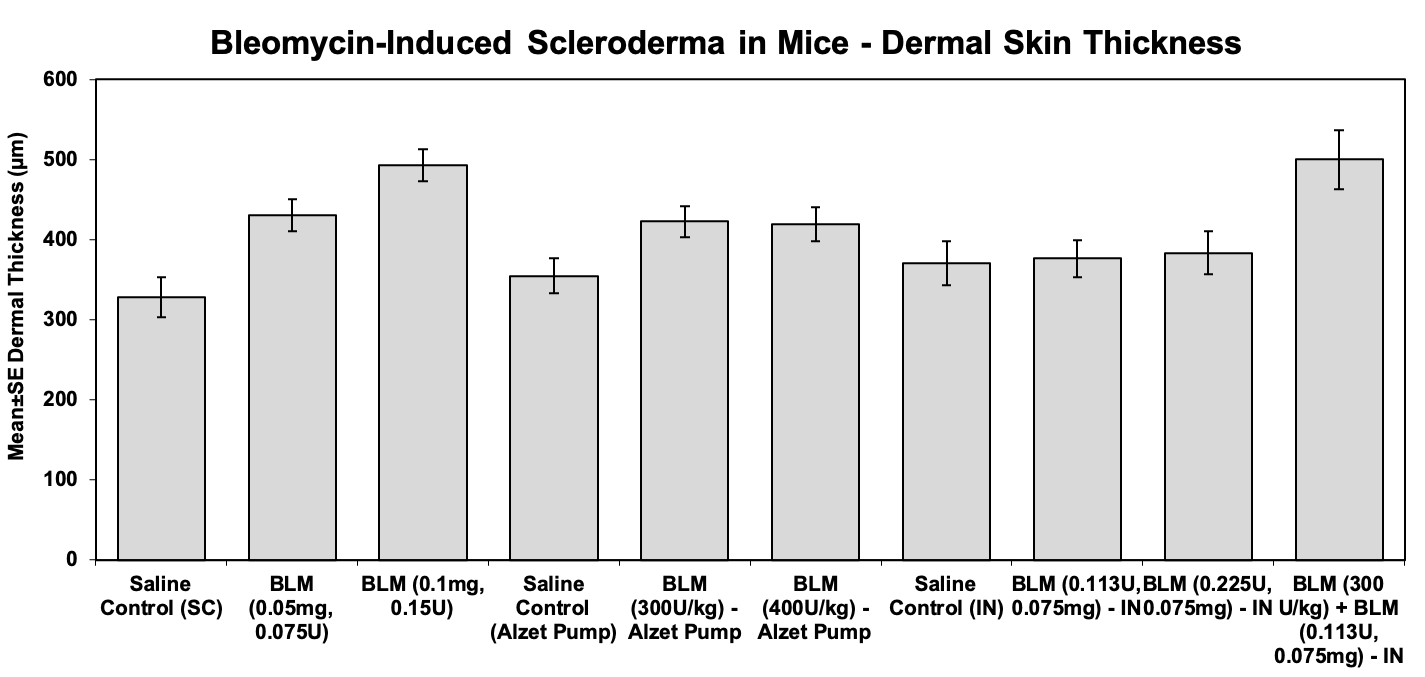
Dermal Thickness Measure (indication of the general magnitude of epidermal thickening)
The skin thickness (dermal-epidermal junction to subcutaneous fat) was measured at 5 sites starting at the left side of each section and progressing to the right side at approximately equal µm intervals. If 2 sections of skin were present, a total of 10 sites were measured.
Dermal Inflammation
- 0=Normal=No inflammation
- 5=Very Minimal, less than 5% of skin affected
- 1=Minimal=5-10% of skin affected
- 2=Mild=11-25% of skin affected
- 3=Moderate=26-50% of skin affected
- 4=Marked=51-75% of skin affected
- 5=Severe=75-100% of skin affected
Lung evaluation
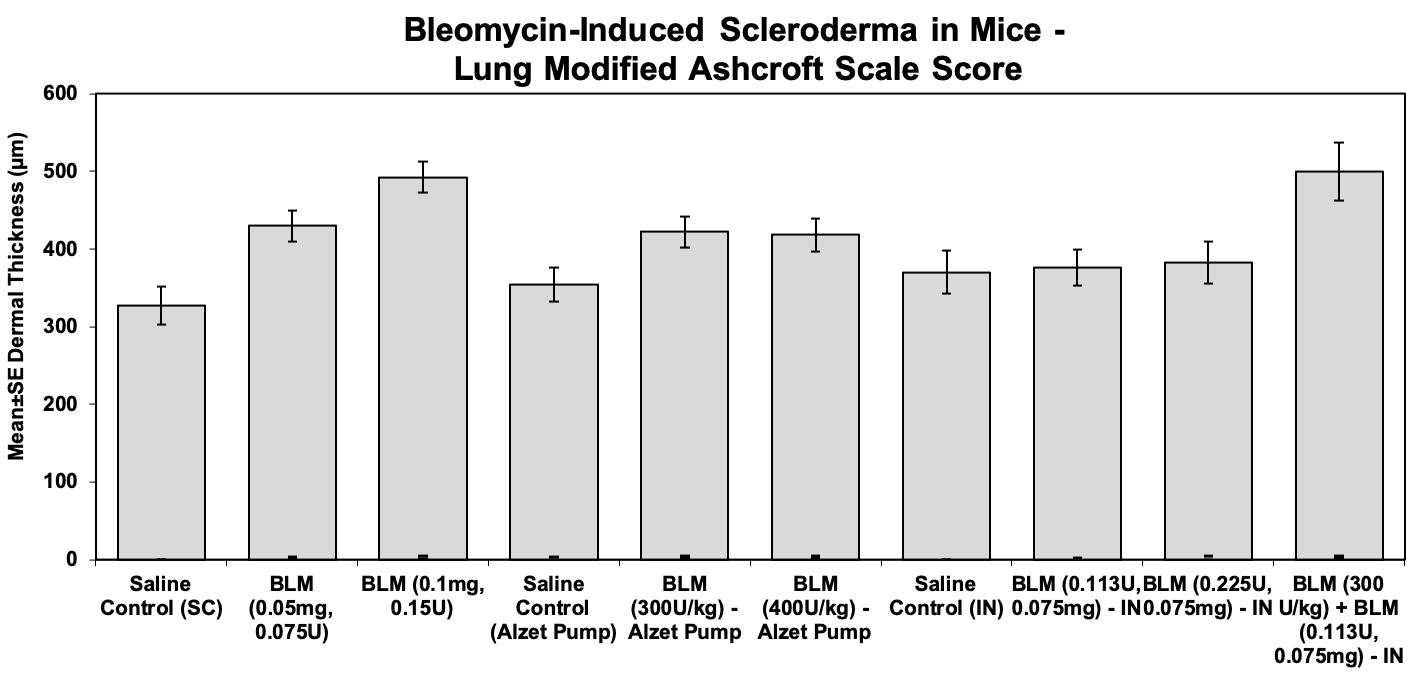
Lung samples are processed and embedded whole, sectioned and stained with H&E and Masson’s Trichrome for pathology evaluation and scoring of fibrosis using the modified Ashcroft Scale.
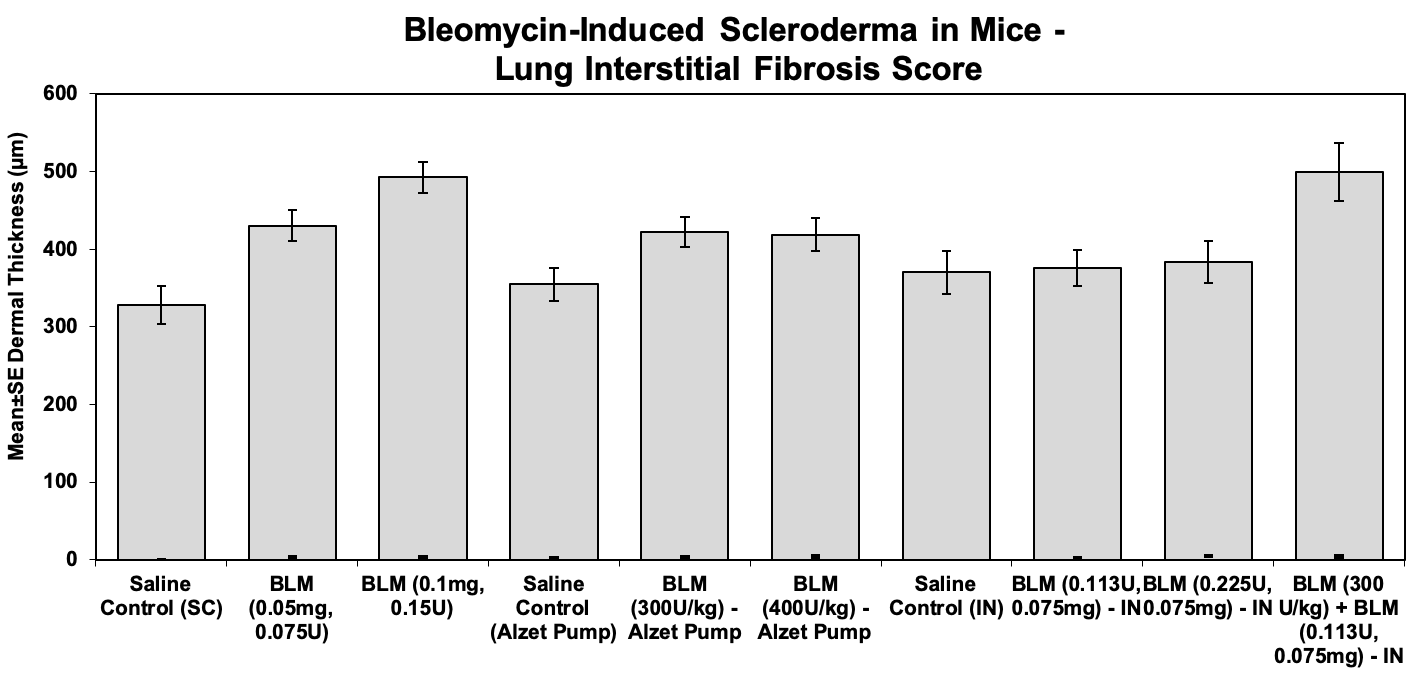
Lungs were scored for the percentage of interstitial fibrosis, as follows:
- 0=None
- 5=Very minimal, <1% of parenchyma affected
- 1=Minimal, 1-5% of parenchyma affected
- 2=Mild, 6-10% of parenchyma affected
- 3=Moderate, 11-15% of parenchyma affected
- 4=Marked, 16-20% of parenchyma affected
- 5=Severe, greater than 20% of parenchyma affected
Modified Ashcroft Scale
Evaluation performed using the 20x objective, with scoring of the most severely affected area as follows:
- 0=Normal
- 1=Isolated alveolar septa with gentle fibrotic changes (septum <3x thicker than normal)
- 2=Fibrotic changes of alveolar septa (>3x thicker than normal) with individual knot-like formations
- 3=Contiguous fibrotic walls of alveolar septa (>3x thicker than normal). No fibrotic masses present.
- 4=Individual fibrotic masses
- 5=Confluent fibrotic masses comprising less than 50% of microscopic field
- 6=Large contiguous fibrotic masses comprising greater than 50% of microscopic field
- 7=Alveolar septa non-existent; alveoli nearly obliterated by fibrous masses, but up to 5 air spaces visible
- 8=Fibrous obliteration of entire microscopic field
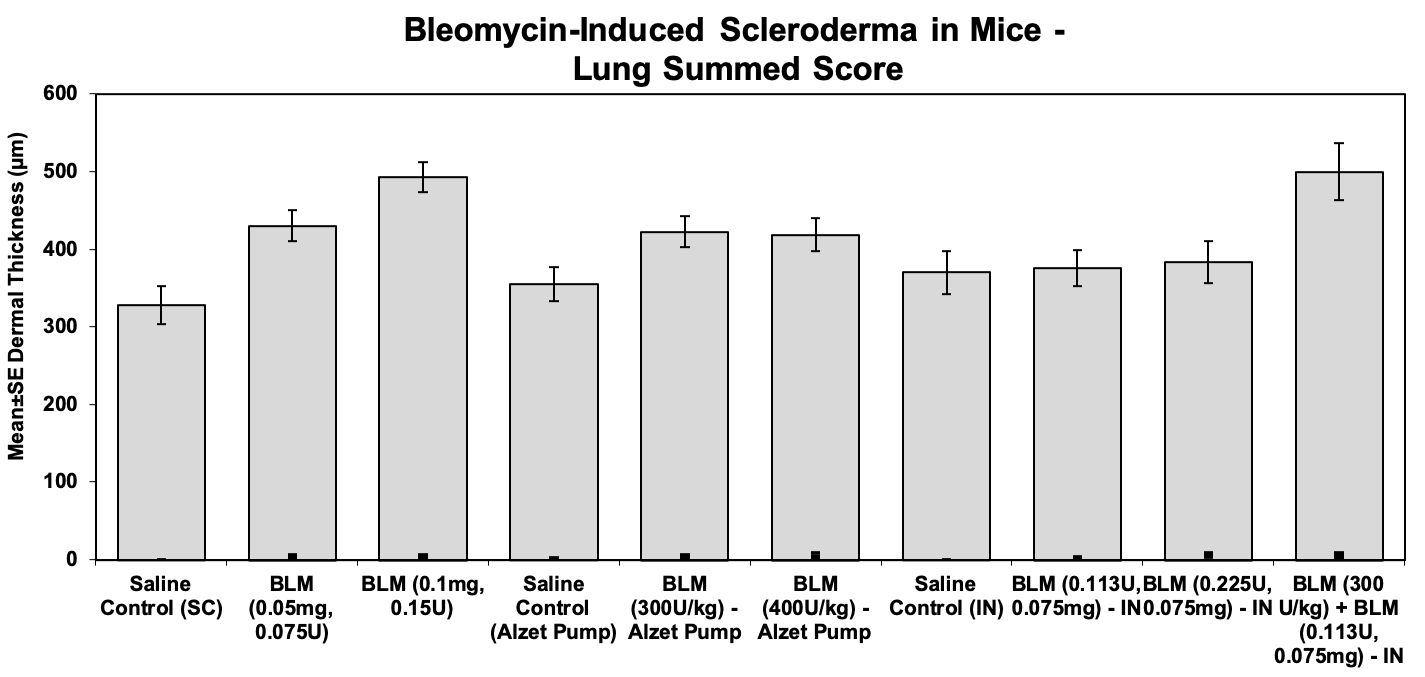
Total lung Score:
Individual scores for percentage of interstitial fibrosis (0-5) and modified Ashcroft scale (0-8) were added to determine the total lung score for each sample.
Sample Data:
References
- Yamamoto T, et al. Animal model of sclerotic skin. I: Local injections of bleomycin induce sclerotic skin mimicking scleroderma. J of Investigative Dermatology. 1999; 112(4): 456- 462.
- Varga J and Abraham D. Systemic sclerosis: a prototypic multisystem fibrotic disorder. J of Clinical Investigation. 2007; 117(3): 557-567.
- Yamamoto T and Kishioka K. Cellular and molecular mechanisms of bleomycin-induced murine scleroderma: current update and future perspective. Exp Dermatol. 2005; 14: 81-95.
- Barnes J and Mayes MD. Epidemiology of systemic sclerosis: incidence, prevalence, survival, risk factors, malignancy, and environmental triggers. Curr Opin Rheumatol. 2012; 24: 165-170.
- Liu S, et al. Role of Rac1 in a bleomycin-induced scleroderma model using fibroblast-specific Rac1-knockout mice. Arthritis & Rheumatism. 2008: 58(7): 2189-2195.
- Servettaz A, et al. Selesctive oxidation of DNA topoisomerase 1 induces systemic sclerosis in the mouse. J of Immunology. 2009; 5855-5864.
- Liang M, et al. Modified murine model of systemic sclerosis: bleomycin given by pump infusion induced skin and pulmonary inflammation and fibrosis. Laboratory Investigation. 2015; 95: 342-350.
- Ishikawa H, et al. Induction of autoimmunity in a bleomycin-induced murine model of experimental systemic sclerosis: an important role for CD4+ T cells. J of Investigative Dermatology. 2009; 129: 1688-1695.
- Tsujino K and Sheppard D. Critical appraisal of the utility and limitations of animal models of scleroderma. Curr Rheumatol Rep. 2016; 18:4.
- Yoshizaki A, et al. CD19 Regulates skin and lung fibrosis via toll-like receptor signaling in a model of bleomycin-induced scleroderma. American Journal of Pathology. 2008; 172(6): 1650-1663.
- Mayes MD, et al. Prevalence, incidence, survival, and disease characteristics of systemic sclerosis in a large US population. Arthritis & Rheumatism. 2003; 48(8): 2246-2255.
Bonifazi M, et al. Systemic sclerosis (scleroderma) and cancer risk: systematic review and meta-analysis of observational studies. Rheumatology. 2013; 52: 143-153.