Glioblastoma Models in Mice

Glioblastoma multiforme (GBM) accounts for 12–15% of all brain tumors and 50–60% of astrocytomas. The frequency of GBM is estimated to be 10 to 100,000 per year, but its average survival time is only ~1 year after diagnosis.1 GBM treatment involves removal of the accessible tumor from the patient’s brain, but its diffuse invasion of the surrounding tissue makes its complete removal difficult. Thus, the likelihood of recurrence is high,2,3 and chemotherapy with paclitaxel (PTX) is essential for the treatment of GBM.4 Unfortunately, the anti-glioma effect of PTX is limited and causes adverse side effects.5,6 Therefore, the development of more effective therapies is necessary.
Induction:
Mice are ordered to be at least 21-42g and at least 4-8 weeks old when on study. Intracranial tumors are established by injecting the rodent-derived C6 glioma cell line into the brain with stereotaxic methods. The skull will be exposed, bregma will be located at the junction of the sagittal and coronal sutures, and the skull will be trephinated 1.8 mm to the right of bregma using a dental drill or similar. The burr hole will be 1-2 mm in diameter, and the syringe needle will then be lowered with the stereotact to 3 mm below bregma, targeting the middle of brain’s striatum. The C6 cells will be injected into the striatum at rate of 2 ml/min, the needle will be withdrawn, and the mouse will be removed from the stereotact. The incision will be sutured closed, and the mouse will be administered 0.05 mg/kg buprenorphine XR (SC; extended release formulation) to mitigate any surgery-related pain. The mouse will then be removed from anesthesia and placed on a heating pad until it regains its righting reflex. The entire procedure is expected to last 10-15 minutes,8-11 and the animals will be maintained and monitored for 2 weeks7 after their surgeries.
As illustrated below, the study design involves all animals receiving tumor induction. One group will receive saline vehicle (IV), while the other will receive PTX (10 mg/kg IV) or test article every other day for 8 days after tumor induction.7
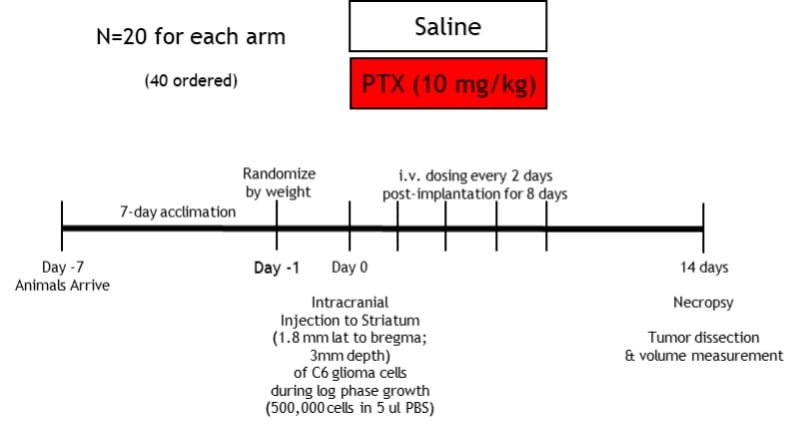
Disease Parameters & Clinical Assessment: On study day 14, all animals will be necropsied and tumor volume will be determined.
Histopathological Assessment: N/A unless specified by client
References
- Iacob, G. and Dinca, E. B. (2009) Current data and strategy in glioblastoma multiforme. J Med Life 2: 386–93.
- Lawrence, R. N. (2002) William Pardridge discusses the lack of BBB research. Drug Discov Today 7: 223–26.
- Pardridge, W. M. (2001) BBB-Genomics: creating new openings for brain-drug targeting. Drug Discov Today 6: 381–83.
- Gupta, M. L., Bode, C. J., Georg, G. I. and Himes, R. H. (2003) Understanding tubulin-Taxol interactions: mutations that impart Taxol binding to yeast tubulin. Proc Natl Acad Sci 100: 6394–97.
- Prados, M. D. et al. (1996) Phase II study of paclitaxel in patients with recurrent malignant glioma. J Clin Oncol 14: 2316–21.
- Chang, S. et al. (2001) A phase II study of paclitaxel in patients with recurrent malignant glioma using different doses depending upon the concomitant use of anticonvulsants: a North American brain tumor consortium report. Cancer 91: 417–22.
- Wang, B. et al (2015) Improved anti-glioblastoma efficacy by IL-13Ra2-mediated copolymer nanoparticles loaded with paclitaxel. Sci Rep 5:16589. doi: 0.1038/srep16589
- Ahlstedt, J., Konradsson, E., Ceberg, C., and Redebrandt, H. N. (2020). Increased effect of two-fraction radiotherapy in conjunction with IDO1 inhibition in experimental glioblastoma. PloS one 15(5): e0233617. doi: 10.1371/journal.pone.0233617
- Habimana-Griffin, L., Ye, D., Carpenter, J., Prior, J., Sudlow, G., Marsala, L., Mixdorf, M., Rubin, J., Chen, H., and Achilefu, S. (2020) Intracranial glioma xenograft model rapidly reestablishes blood-brain barrier integrity for longitudinal imaging of tumor progression using fluorescence molecular tomography and contrast agents. J Biomed Opt 25(2): 1–13. doi: 10.1117/1.JBO.25.2.026004
- Lee, M., Muller, F., Aquilanti, E., Hu, B., and DePinho, R. (2012) Stereotactic orthotopic xenograft injections into the mouse brain. Protocol Exchange doi:10.1038/protex.2012.041.
- Jarry, U., Joalland, N., Chauvin, C., Clemenceau, B., Pecqueur, C., and Scotet, E. (2018). Stereotactic adoptive transfer of cytotoxic immune cells in murine models of orthotopic human glioblastoma multiforme xenografts. J Vis Exp 139: 57870. https://doi.org/10.3791/57870
Related Pages
- Glioblastoma Models in Mice
- Epilepsy Models in Rats and Mice
- Addiction Models in Rats and Mice
- Cognition Models in Rats and Mice
- Experimental Autoimmune Encephalomyelitis (EAE)
- Developmental Milestones Aka Functional Observation Battery
- Anxiety Models in Rats and Mice
- Depression Model in Rat
- Fragile X Syndrome (FXS) in Mice
- Parkinson’s Disease Models in Rats and Mice
- Alzheimer’s Disease Model in Mice

